
Hydrogen bohr model bossguide
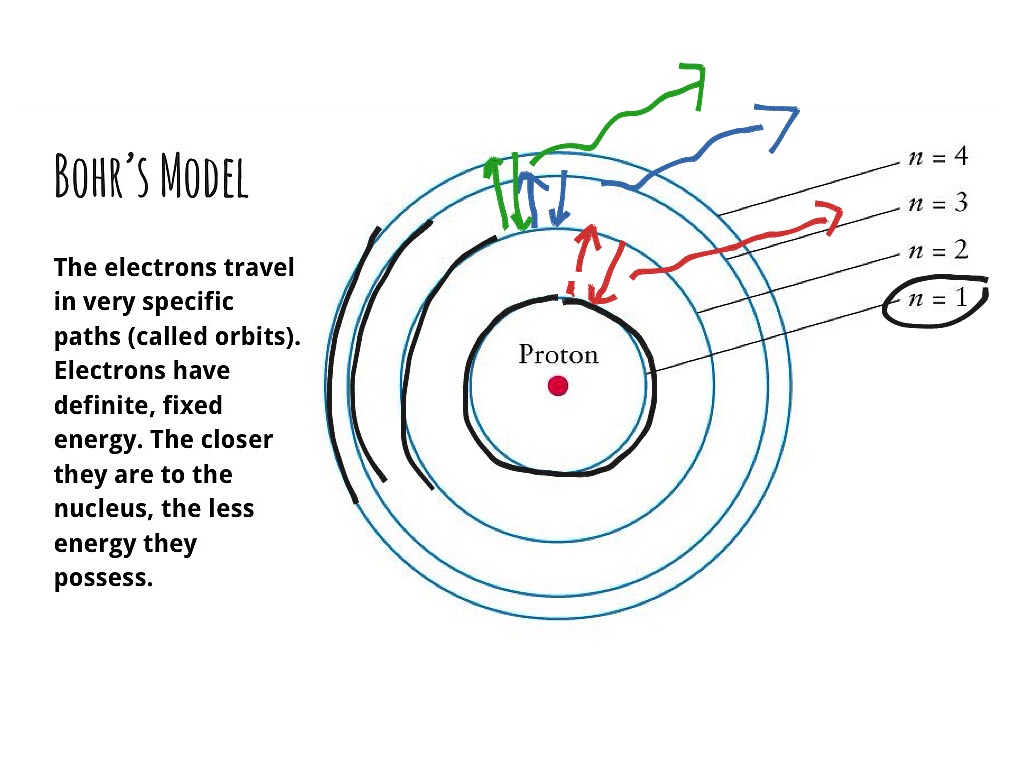
The simplest example of the Bohr Model is for the hydrogen atom (Z = 1) or for a hydrogen-like ion (Z > 1), in which a negatively charged electron orbits a small positively charged nucleus. Electromagnetic energy will be absorbed or emitted if an electron moves from one orbit to another. Only certain electron orbits are permitted.
Bohr Model Model Atomic Iron Atomic Orbital PNG, Clipart, Area, Atom
Since the Rydberg constant was one of the most precisely measured constants at that time, this level of agreement was astonishing and meant that Bohr's model was taken seriously, despite the many assumptions that Bohr needed to derive it. Figure \(\PageIndex{1}\): Quantum numbers and energy levels in a hydrogen atom.

Bohr model Atomic nucleus Atomic theory Iron, atomic, chemical Element
The Bohr model, introduced by Danish physicist Niels Bohr in 1913, was a key step on the journey to understand atoms. Ancient Greek thinkers already believed that matter was composed of tiny.

Iron atom Bohr model stock vector. Illustration of isolated 267662069
Immediately before 1913, the Rutherford model conceived of an atom as consisting of a tiny positively charged heavy core, called a nucleus, surrounded by light, planetary negative electrons revolving in circular orbits of arbitrary radii. Britannica Quiz Matter and More Quiz How does Niels Bohr's atomic model work?
3d render of atom structure of iron Stock Photo by ©oorka5 136542118
Bohr diagrams show electrons orbiting the nucleus of an atom somewhat like planets orbit around the sun. In the Bohr model, electrons are pictured as traveling in circles at different shells, depending on which element you have. Figure 2 2 contrast the Bohr diagrams for lithium, fluorine and aluminum atoms. The shell closest to the nucleus is.

[DIAGRAM] Frost Diagram For Aluminum
Bohr's model suggests that the atomic spectra of atoms is produced by electrons gaining energy from some source, jumping up to a higher energy level, then immediately dropping back to a lower energy level and emitting the energy difference between the two energy levels. The existence of the atomic spectra is support for Bohr's model of the atom.

5 Cool 3d Model Of Silicon Atom Wanted Mockup
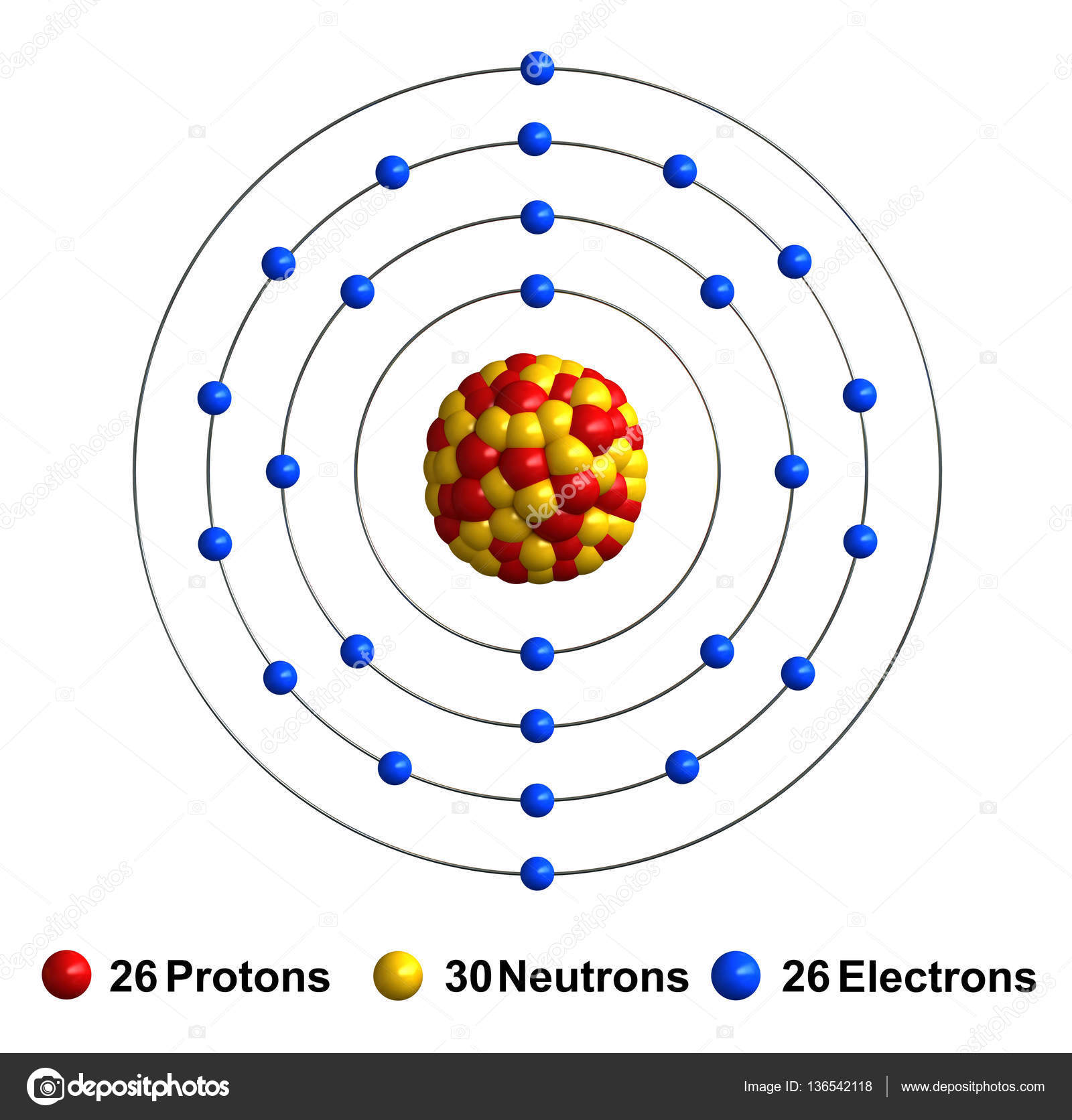
Basic Information Name: Iron Symbol: Fe Atomic Number: 26 Atomic Mass: 55.845 amu Melting Point: 1535.0 °C (1808.15 K, 2795.0 °F) Boiling Point: 2750.0 °C (3023.15 K, 4982.0 °F) Number of Protons/Electrons: 26 Number of Neutrons: 30 Classification: Transition Metal Crystal Structure: Cubic Density @ 293 K: 7.86 g/cm 3 Color: Silvery

Iron(III) Chloride AMERICAN ELEMENTS ® / Ferric Chloride
Bohr's model suggests that each atom has a set of unchangeable energy levels, and electrons in the electron cloud of that atom must be in one of those energy levels.. The image below shows the emission spectrum of iron. Because each element has a unique emission spectrum, elements can be defined using them. Figure \(\PageIndex{3}\): Atomic.

Periodic Network 2012 [licensed for use only] / Iron
The electron cloud model An electron cloud model of a helium-4 atom is shown below. [What do the scales mean on this model?] In this model, the black "cloud" represents the volume of space where electrons are likely to be found. The darker the region, the more likely electrons are to be found there.
Iron (Fe) AMERICAN ELEMENTS
The Bohr Model is a modification of an earlier atomic model, the Rutherford Model. The Bohr Model has an atom with a positively-charged nucleus surrounded by negatively-charged electrons that have circular, planetary-like orbits. Today, we know that the Bohr Model has some inaccuracies, but it's still used because of its simple approach to.
Bohr Model of the Atom Overview and Examples
The Bohr model of the hydrogen atom (Z = 1) or a hydrogen-like ion (Z > 1), where the negatively charged electron confined to an atomic shell encircles a small, positively charged atomic nucleus and where an electron jumps between orbits, is accompanied by an emitted or absorbed amount of electromagnetic energy (hν). The orbits in which the electron may travel are shown as grey circles; their.
ShowMe bohr model for carbon
The Bohr Model for Hydrogen (and other one-electron systems) In 1913, a Danish physicist, Niels Bohr (1885-1962; Nobel Prize in Physics, 1922), proposed a theoretical model for the hydrogen atom that explained its emission spectrum.. iron, and carbon, along with smaller amounts of other elements. During the solar eclipse of 1868, the.

Arriba 63+ imagen hierro modelo de bohr Abzlocal.mx
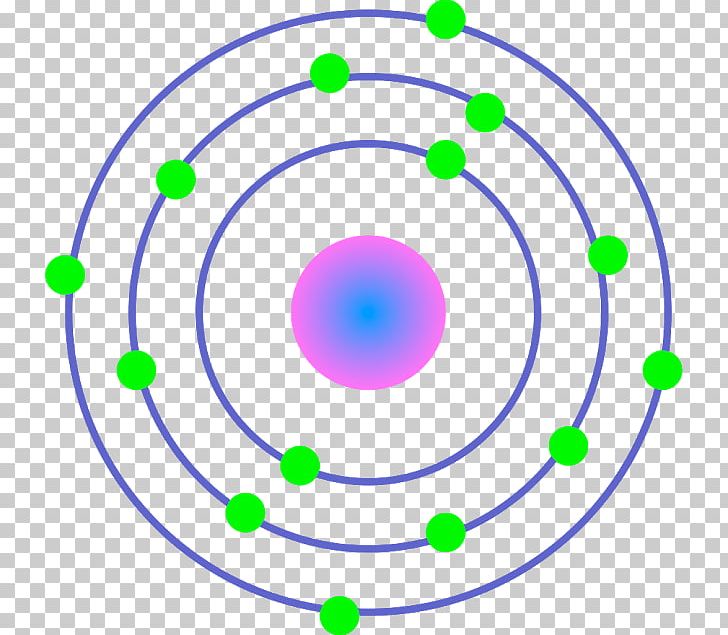
The Bohr Model of Iron (Fe) has a nucleus that contains 30 neutrons and 26 protons. This nucleus is surrounded by four electron shells namely K-shell, L-shell, M-shell, and N-shell. The 1 st shell has 2 electrons, the 2 nd shell has 8 electrons, the 3 rd shell has 14 electrons and the 4 th shell has 2 electrons. Page Contents show

Iron Atom Structure Stock Vector Art & More Images of 2015 493163650
The Bohr model describes the structure of an atom as a central nucleus containing protons and neutrons, with electrons orbiting in specific energy levels around it. Electrons can jump between these energy levels by absorbing or emitting energy. This model helps us understand basic atomic properties and electron behavior in atoms. K Shell

Iron, atomic structure Stock Image C018/3707 Science Photo Library
Niels Henrik Bohr (1885-1962) was a Danish physicist who developed the atomic model and won the 1922 Nobel prize in physics. Photo 12/Getty Images "Bohr's model of 1913 for the hydrogen atom had circular electron orbits about the proton — like Earth orbits around the sun," says Herschbach.
:max_bytes(150000):strip_icc()/Iron-58b602243df78cdcd83d3d5a.jpg)
Iron Periodic Table Electrons Review Home Decor
Bohr's model consists of a small nucleus (positively charged) surrounded by negative electrons moving around the nucleus in orbits. Bohr found that an electron located away from the nucleus has more energy, and the electron which is closer to nucleus has less energy Postulates of Bohr's Model of an Atom